 .svg/500px-Periodic_table_(polyatomic).svg.png)
These atoms are often called the alkali and alkaline earth elements. For example, potassium has a configuration Ar4s 1. 
1 ), the configuration is a closed shell of core electrons, plus s electrons in a new shell. In other words, the number of valence electrons for a transition metal is equal to how many spaces across the periodic table it is. For atoms found in the first two columns of the periodic table (figure 1.6.1 1.6. The valence electrons for transition metals are equal to the number of s-electrons plus the number of d-electrons. For non-transition metals, we count to 8, but for transition metals, we count to 12. Looking at the orbitals explains how valence electrons work for transition metals. For example, oxygen has 6 valence electrons, these six electrons fill up the 2s orbital, and partially fill the 2p subshells (2s 22p 4). The shells after (ignoring transition metals) represent the s and p-orbitals. So what does this have to do with our shells? The first "shell" represents the 1s orbital. For example, carbon has an electron configuration of 1s 22s 22p 2. The way we count our electrons is by moving from right to left, starting at the beginning of the table. F-orbitals start appearing in the lanthanides and actinides (the separated two rows). P-orbitals start appearing in period 2, and d-orbitals start appearing in period 4 (though they start counting at 3). Has 7 subshells, each holding 2 electrons, for a total of 14 electronsīelow is the periodic table with the labeled orbitalsĮach period is its own energy level. 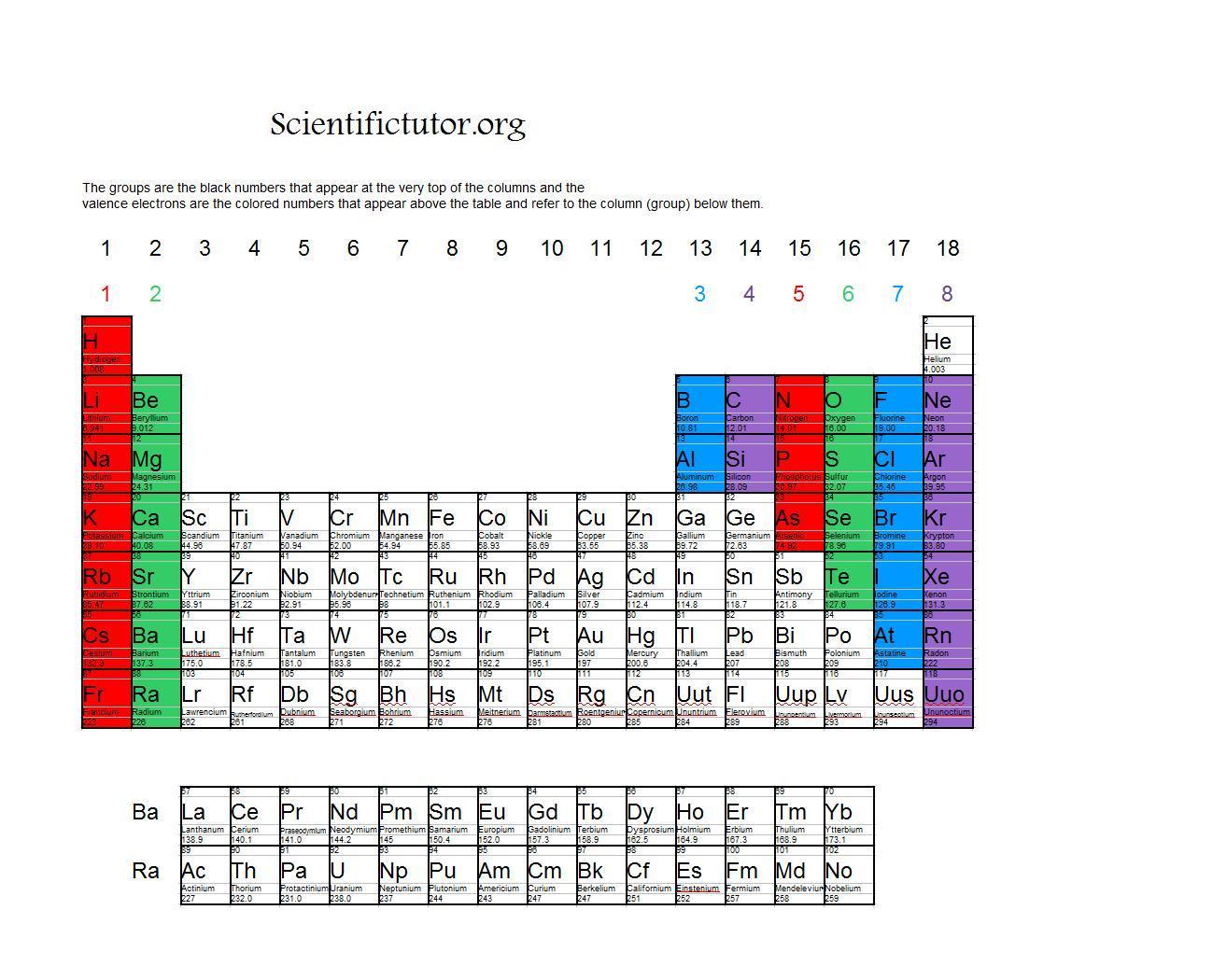
Has 5 subshells, each holding 2 electrons, for a total of 10 electrons.Has 3 subshells, each holding 2 electrons, for a total of 6 electrons.The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons. There are total 18 vertical columns on periodic table. The Arabic numbering system is the most widely accepted today. Groups are the vertical columns on the periodic table. Many periodic tables include both Roman and Arabic numbers. The modern IUPAC system uses Arabic numbers 1-18, simply numbering the columns of the periodic table from left to right.The CAS system used letters to differentiate main group (A) and transition (B) elements.The older IUPAC system used Roman numerals together with letters to distinguish between the left (A) and right (B) side of the periodic table.Three systems have been used to number families and groups: Recognizing Families on the Periodic TableĬolumns of the periodic table typically mark groups or families. 
Noble Gases: - Group 18 (VIIIA) - 8 valence electrons.Halogens: - Group 17 (VIIA) - 7 valence electrons.Oxygen Group or Chalcogens: - Group 16 (VIA) - 6 valence electrons.Nitrogen Group or Pnictogens: - Group 15 (VA) - 5 valence electrons.Carbon Group or Tetrels: - Group 14 (IVA) - 4 valence electrons.Boron Group or Earth Metals: Group 13 (IIIA) - 3 valence electrons.Transition Metals: Groups 3-12 - d and f block metals have 2 valence electrons.Alkaline Earth Metals: Group 2 (IIA) - 2 valence electrons.Alkali Metals: Group 1 (IA) - 1 valence electron.Many chemists and chemistry textbooks recognize five main families:Īnother common method of categorization recognizes nine element families: However, there are different ways of categorizing elements into families. Because element properties are largely determined by the behavior of valence electrons, families and groups may be the same. Now, for the sake of this video, Im gonna focus most on the extremes of the periodic table, the groups at the left and the right. Since the number of valence electrons is a periodic property, the value can be easily determined by locating the element in the periodic table. Element groups, on the other hand, are collections of elements categorized according to similar properties. And because elements with similar valence electrons will have similar reactivities, they will form similar ions. We can write the configuration of oxygen's valence electrons as 2s2p. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell. The characteristics of the elements in these families are determined primarily by the number of electrons in the outer energy shell. The entire text of Teaching Valence Electrons and the Periodic Table video with embedded questions aligned to Common Core and Depth of Knowledge (DOK). Transcript Valence electrons are the electrons in the outermost shell, or energy level, of an atom. Elements are classified into families because the three main categories of elements (metals, nonmetals, and semimetals) are very broad. Element families are indicated by numbers located at the top of the periodic table.Īn element family is a set of elements sharing common properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
